Chinese firm gets approval to begin human testing for potential coronavirus vaccine
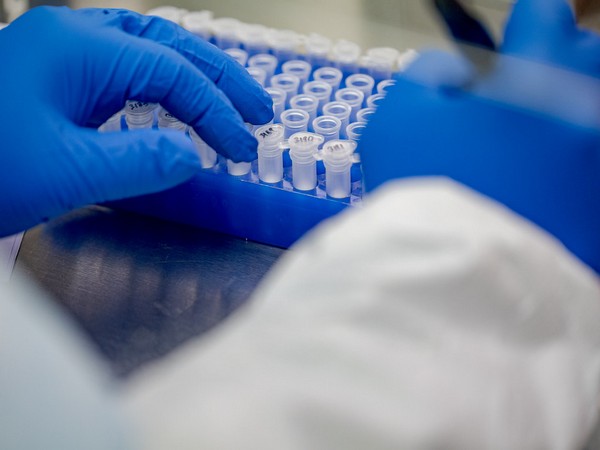
The potential vaccine, co-developed by Anhui Zhifei Longcom Biopharmaceutical and the Institute of Microbiology of the Chinese Academy of Sciences, has received a certificate from the National Medical Products Administration to launch clinical trials. Chinese researchers and companies are testing six experimental shots in humans, and more than a dozen vaccines are in different stages of clinical trials globally against the virus that has killed over 470,000 people.
- Country:
- China
China has approved a coronavirus vaccine candidate developed by Chongqing Zhifei Biological Products' unit to begin human testing, the company said in a filing on Tuesday. The potential vaccine, co-developed by Anhui Zhifei Longcom Biopharmaceutical and the Institute of Microbiology of the Chinese Academy of Sciences, has received a certificate from the National Medical Products Administration to launch clinical trials.
Chinese researchers and companies are testing six experimental shots in humans, and more than a dozen vaccines are in different stages of clinical trials globally against the virus that has killed over 470,000 people. However, none of them have passed large-scale, late-stage phase 3 clinical trials, a necessary step before entering the consumer market.
(This story has not been edited by Devdiscourse staff and is auto-generated from a syndicated feed.)
ALSO READ
Between the US and a hard place, Germany's Scholz reheats China ties
China commerce ministry says Envalior to inherit 8.2% anti-dumping tax
Former China Everbright Group chairman charged with taking bribes, state media reports
China, Japan coast guards patrol disputed East China Sea waters
China launch of relay satellite Queqiao-2 for lunar probe mission successful










