UPDATE 1-Biotech company Novacyt seeks emergency approval for coronavirus test
- Country:
- United States
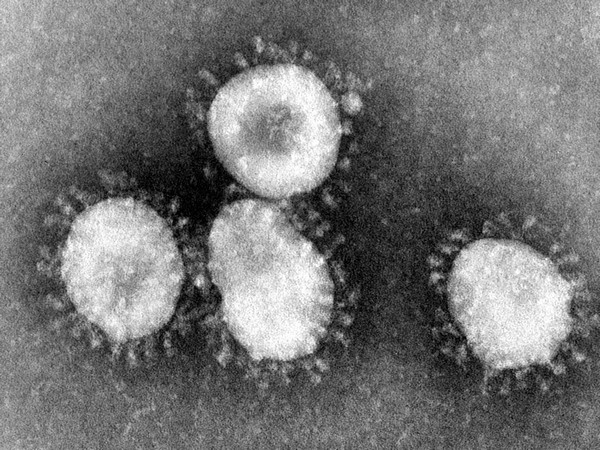
Biotechnology company Novacyt has applied for an emergency approval from the U.S. Food & Drug Administration (FDA) as it races to roll out a product that tests for the coronavirus, the company said on Friday.
Novacyt, which is listed in Paris and London, also said it was in talks with public sector hospitals in Britain. Its Paris-listed shares jumped by around 20 percent by 0830 GMT.
"As one of the first companies to develop and launch a test to detect the 2019 strain of nCoV (coronavirus), we have received unprecedented interest," Novacyt CEO Graham Mullis said. "We continue to support our new and existing customers and are working with the various regulatory authorities to try to make our nCoV test available to as many countries and laboratories as possible," he added.
The death toll from the fast-spreading coronavirus in mainland China has reached 636, with 73 more deaths recorded as of Thursday, China's National Health Commission said.
(This story has not been edited by Devdiscourse staff and is auto-generated from a syndicated feed.)
- READ MORE ON:
- Food & Drug Administration
- Paris
- London
- Britain
- China
- National Health Commission
ALSO READ
India ready for Australia challenge ahead of Paris Olympics
London stocks fall 1% as geopolitical tensions and economic data loom
India ready for Australia challenge ahead of Paris Olympics
Elgar Parishad-Maoist links case: SC grants bail to activist Shoma Kanti Sen
SC grants bail to activist Shoma Kanti Sen in Elgar Parishad-Maoist links case.










