EU regulator recommends dose gap of 3 weeks for Pfizer COVID-19 vaccine
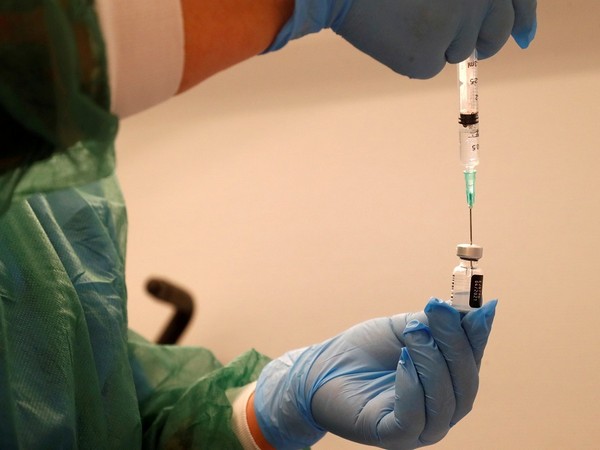
Europe's drugs regulator said on Thursday it now recommended that the second dose of Pfizer's COVID-19 vaccine be given three weeks after the first as many countries delay shots to vaccinate more of their population.
The labelling of the vaccine, developed along with Germany's BioNTech and branded Comirnaty in Europe, before the change stated that the interval between the two doses should be "at least 21 days", the European Medicines Agency said. (https://bit.ly/3ciKHiB)
(This story has not been edited by Devdiscourse staff and is auto-generated from a syndicated feed.)
- READ MORE ON:
- Pfizer
- European Medicines Agency
- Europe
- Germany
Advertisement
ALSO READ
Pfizer RSV shot meets goals in trial of high-risk adults under age 60
Health News Roundup: Pfizer RSV shot meets goals in trial of high-risk adults under age 60; Novartis to cut 680 jobs in product development and more
Health News Roundup: Pfizer RSV shot meets goals in trial of high-risk adults under age 60; Cuba says drug use on the rise, especially among youth and more










