Health News Roundup: Factbox-Monkeypox cases around the world; U.S. drug regulator lifts clinical hold on Ocugen's COVID vaccine trial and more
The Food and Drug Administration paused the trials of the shot, Covaxin, in April after an inspection of a Bharat Biotech facility by the World Health Organization (WHO) revealed deficiencies in the manufacturing process. Third possible case of monkeypox found in the U.S Health authorities said they may have found a third case of the monkeypox virus in the United States and are running tests on a patient in South Florida to confirm if the person has contracted the disease, which is staging a rare outbreak outside of Africa.
Following is a summary of current health news briefs.
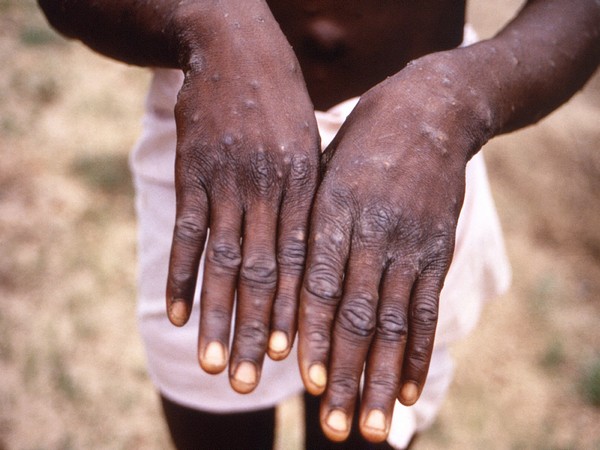
Factbox-Monkeypox cases around the world
Nearly 20 countries where monkeypox is not endemic have reported outbreaks of the viral disease, with more than 100 confirmed or suspected infections mostly in Europe. The outbreaks are raising alarm because monkeypox, which spreads through close contact and was first found in monkeys, mostly occurs in the west and central Africa, and only very occasionally spreads elsewhere.
U.S. drug regulator lifts clinical hold on Ocugen's COVID vaccine trial
Ocugen Inc said on Monday the U.S. drug regulator has lifted the clinical hold on a mid-to-late stage trial of the COVID-19 vaccine being developed by its Indian partner Bharat Biotech. The Food and Drug Administration paused the trials of the shot, Covaxin, in April after an inspection of a Bharat Biotech facility by the World Health Organization (WHO) revealed deficiencies in the manufacturing process.
Third possible case of monkeypox found in the U.S
Health authorities said they may have found a third case of the monkeypox virus in the United States and are running tests on a patient in South Florida to confirm if the person has contracted the disease, which is staging a rare outbreak outside of Africa. The case in Broward County, Florida, is "related to international travel," the U.S. Centers for Disease Control and the Florida Department of Health said in a statement on Sunday, "and the person remains isolated."
AstraZeneca says EU regulator approves COVID shot as booster
Drugmaker AstraZeneca said on Monday its COVID-19 vaccine, Vaxzevria, has been approved in the European Union by the bloc's drugs regulator as a third-dose booster in adults following a committee endorsement last week. The vaccine can now be used as a booster following the two-dose Vaxzevria schedule or by those who have been previously vaccinated by an mRNA vaccine, such as the ones made by Pfizer and BioNTech or Moderna.
Biohaven's drug for neurological disease fails late-stage study
Biohaven Pharmaceutical said on Monday its experimental drug for patients with spinocerebellar ataxia, a genetic disease that affects the nervous system, failed to meet the main goal of a late-stage study. Spinocerebellar ataxia (SCA) is a progressive disorder caused by the degeneration of cells in the brain and spinal cord and can lead to symptoms such as uncoordinated movement and muscle wasting. It has no approved treatments in the United States.
U.S. Task Force to consider routine kidney disease screening
An influential U.S. panel has added screening for chronic kidney disease (CKD) to its list of preventive services under active consideration, the group's chair told Reuters, a move that could help identify patients eligible for new drugs that treat the disease earlier. The U.S. Preventive Services Task Force (USPSTF) last addressed the issue in 2012, finding insufficient evidence to assess routine screening for CKD in asymptomatic adults. That conclusion does not reflect the latest science and is now inactive, Dr. Carol Mangione, chair of the government-backed panel, said in an email.
Pfizer/BioNTech say 3 COVID shots generate good response in under-5s
Drugmakers Pfizer Inc and BioNTech SE said on Monday that three doses of their COVID-19 vaccine generated a strong immune response in children under the age of five in their clinical trial. The companies said the vaccine was safe and well-tolerated by the children, and they plan to soon ask global regulators to authorize the shot for the age group, children for whom no vaccine is currently approved in most of the world.
Beijing extends work-from-home 'requirement' for millions as COVID spreads
The Chinese capital extended its work-from-home requirement for many of its 22 million residents to stem a COVID-19 outbreak, while Shanghai deployed more testing and curbs to hold on to its hard-won "zero COVID" status after two months of lockdown. Beijing said 99 new cases were detected on Sunday, up from 61 the previous day - the largest daily tally so far during a month-old outbreak that has consistently seen dozens of new infections every day.
Sri Lankan medicine shortage a death sentence for some, doctors say
A shortage of medicine caused by an economic crisis in Sri Lanka could soon cause deaths, doctors said, as hospitals are forced to postpone life-saving procedures for their patients because they do not have the necessary drugs. Sri Lanka imports more than 80% of its medical supplies but with foreign currency reserves running out because of the crisis, essential medications are disappearing from shelves and the healthcare system is close to collapse.
UK monitoring cases of monkeypox as Scotland detects first infection
Prime Minister Boris Johnson said on Monday the government was looking carefully at the circumstances surrounding the transmission of monkeypox in Britain as Scotland confirmed its first case of the viral infection. "It is basically a very rare disease and so far the consequences don’t seem to be very serious but it is important that we keep an eye on it," British Prime Minister Boris Johnson told reporters.










