Health News Roundup: U.S. CDC expands use of bivalent COVID vaccines for kids as young as 6 months; Patient selection for AstraZeneca, Daiichi breast cancer drug needs improvement, experts say and more
The World Health Organization (WHO) and the U.S. Food and Drug Administration have both gone a step further and pulled recommendations for some monoclonal antibodies on the basis that Omicron and the variant's latest offshoots have likely rendered these options obsolete. Juul agrees to pay $1.2 billion in youth-vaping settlement - Bloomberg News Juul Labs Inc has agreed to pay $1.2 billion to resolve about 10,000 lawsuits targeting the e-cigarette maker as a major cause of a U.S. youth-vaping epidemic, Bloomberg News reported on Friday, citing people familiar with the matter.
Following is a summary of current health news briefs.
U.S. CDC expands use of bivalent COVID vaccines for kids as young as 6 months
The U.S. Centers for Disease Control and Prevention (CDC) on Friday expanded the use of COVID-19 vaccines that target both the original coronavirus and Omicron sub-variants to include children aged 6 months through 5 years. The development comes a day after the U.S. Food and Drug Administration authorized the updated shots from Moderna as well as Pfizer and its partner BioNTech for use in children as young as 6 months.
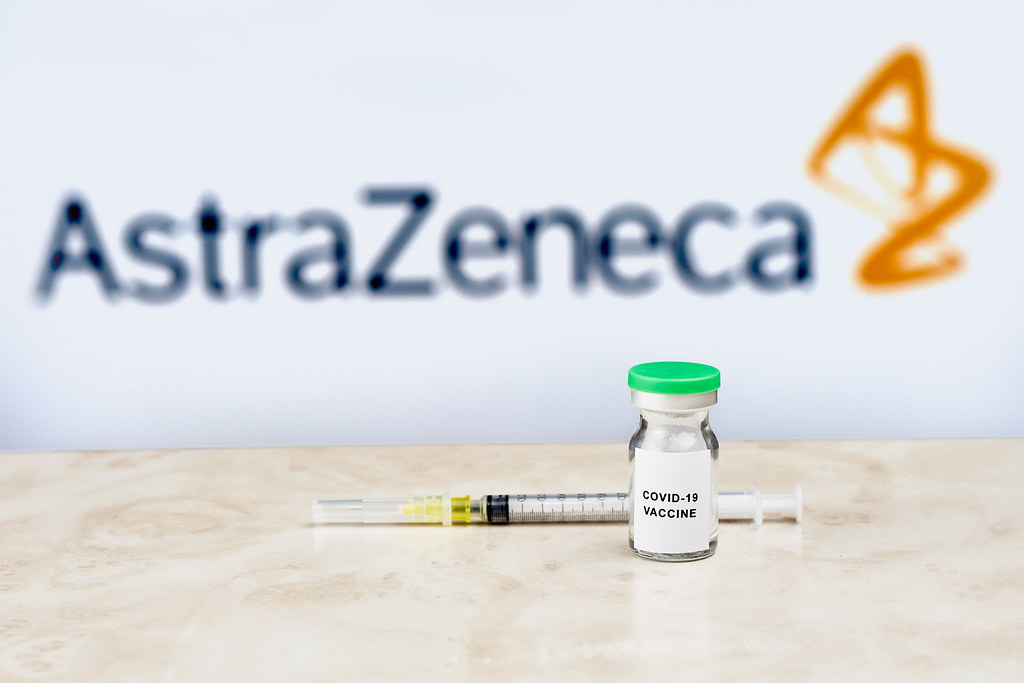
Patient selection for AstraZeneca, Daiichi breast cancer drug needs improvement, experts say
The rush to use AstraZeneca and Daiichi-Sankyo's drug Enhertu to treat certain types of breast cancer has far outpaced doctors' ability to determine with certainty which patients might benefit, experts said this week at a meeting of breast cancer doctors. Enhertu, which won U.S. approval in late 2019, is used in patients with advanced breast, gastric and lung cancers whose tumor cells carry a protein called HER2.
'It's dead out here': China's slow exit from zero-COVID
Judging by Friday's quiet streets in China's capital Beijing and the reluctance of some businesses to drop COVID curbs, enduring anxieties about the coronavirus are likely to hamper a speedy return to health for the world's second-largest economy. Although the government on Wednesday loosened key parts of its strict "zero-COVID" policy that has kept the pandemic largely at bay for the past three years, many people appear wary of being too quick to shake off the shackles.
China to allow German expats to use German COVID-19 vaccines
The Chinese foreign ministry said China and Germany had reached an agreement on providing "German vaccines" to German nationals in China, after the German Chancellor recently said that BioNTech's COVID-19 vaccine would be used by German expatriates. Relevant arrangements will be discussed and determined by the two sides through diplomatic channels, Mao Ning, a spokeswoman at the Chinese foreign ministry told reporters on Friday at a regular press conference.
Some bloodstream infection bacteria grew resistant to last-resort drugs in 2020 - WHO
Increased drug resistance in bacteria causing bloodstream infections, including against last-resort antibiotics, was seen in the first year of the coronavirus pandemic, a World Health Organization report based on data from 87 countries in 2020 showed. The overuse and/or misuse of antibiotics has helped microbes to become resistant to many treatments, while the pipeline of replacement therapies in development is alarmingly sparse.
White House doctors urge Americans to get updated COVID boosters
Top U.S. health officials on Friday urged Americans to get COVID-19 vaccine boosters if eligible to help ward off infections during the holiday season. Speaking at a virtual town hall, White House COVID-19 Response Coordinator Dr. Ashish Jha suggested people who had been infected with COVID in September or earlier consider getting an anti-Omicron booster shot.
FDA staff reviewers flag safety concerns over Cytokinetics' heart drug
U.S. Food and Drug Administration staff reviewers on Friday identified safety and efficacy concerns about Cytokinetics Inc's heart drug, according to briefing documents published on the agency's website. The late-stage data raises concerns about the drug's dose-limiting risks of heart failure, the reviewers said, adding it is unclear if the study would be enough to provide evidence of the treatment's effectiveness.
EU watchdog warns on waning effectiveness of antibody COVID drugs
The European Union's health regulator on Friday raised concerns over the decreasing effectiveness of COVID-19 antibody therapies to treat patients as emerging virus variants are challenging treatment approaches. The World Health Organization (WHO) and the U.S. Food and Drug Administration have both gone a step further and pulled recommendations for some monoclonal antibodies on the basis that Omicron and the variant's latest offshoots have likely rendered these options obsolete.
Juul agrees to pay $1.2 billion in youth-vaping settlement - Bloomberg News
Juul Labs Inc has agreed to pay $1.2 billion to resolve about 10,000 lawsuits targeting the e-cigarette maker as a major cause of a U.S. youth-vaping epidemic, Bloomberg News reported on Friday, citing people familiar with the matter. Last week, Juul said it had settlements with about 10,000 plaintiffs covering more than 5,000 cases in California. The company chose not to disclose the settlement amount as part of the court process in the federal multi-district litigation.
China cracks down on drug price gouging amid fears of COVID spike
Chinese authorities said they were cracking down on price gouging of medical supplies and drugs, and seeking to ensure they flow smoothly as fears grow of a massive COVID-19 outbreak after the nationwide dismantling of strict restrictions. China announced 10 measures on Wednesday that loosened key parts of President Xi Jinping's signature zero-COVID policy, in a dramatic pivot toward economic reopening. But concerns are increasing of a spike in infections as people scramble for cough medicines, flu drugs and masks.
(With inputs from agencies.)
ALSO READ
Germany to Release Russian Bank Assets in Support of Ukraine
Financial Moves: New Regulations, Strategic Listings, and Germany's Investment Push
Germany's Extremist Youth: An Unfolding Threat to Democracy
Germany Urges Caution Over U.S. Venezuela Oil Blockade
Germany Renews Partnership with UNDP to Strengthen Sustainable Development Efforts










