Health News Roundup: UK designates monkeypox as a notifiable disease; Whistleblower sues Eli Lilly over drug manufacturing problems and more
Following is a summary of current health news briefs.
U.S. CDC removes mask recommendation from monkeypox travel notice to avoid confusion
The U.S. Centers for Disease Control and Prevention (CDC) said on Tuesday it had removed a mask recommendation from its monkeypox travel notice to avoid "confusion" over the disease, which primarily spreads through direct contact. "Late yesterday, CDC removed the mask recommendation from the monkeypox Travel Health Notice because it caused confusion," a CDC spokesperson said on Tuesday.
Food shortages are next global health crisis - expert
Growing food shortages may represent the same health threat to the world as the COVID-19 pandemic, a leading global health figure has warned. Rising food and energy prices, in part sparked by the war in Ukraine, could kill millions both directly and indirectly, Peter Sands, the executive director of the Global Fund to Fight AIDS, Tuberculosis and Malaria, told Reuters in an interview on Tuesday.
Omicron sub-variants BA.4 and BA.5 account for up to 13% of COVID variants in U.S. - CDC
The BA.4 and BA.5 sub-variants of Omicron are estimated to make up nearly 5% and 8% of the coronavirus variants in the United States as of June 4, the U.S. Centers for Disease Control and Prevention (CDC) said on Tuesday. The two sublineages, which were added to the World Health Organization's monitoring list in March and designated as variants of concern by the European Centre for Disease Prevention and Control (ECDC), were present in all U.S. regions.
Whistleblower sues Eli Lilly over drug manufacturing problems
A former human resources officer at Eli Lilly and Co sued the drugmaker on Monday, alleging she was terminated after pointing out poor manufacturing practices and data falsification involving one of its blockbuster diabetes drugs. The plaintiff, Amrit Mula, alleges violations of an employee protection law and seeks unspecified damages. The U.S. Department of Justice last year launched a criminal investigation into alleged manufacturing irregularities at an Eli Lilly plant in New Jersey following a Reuters story that detailed some of Mula's allegations.
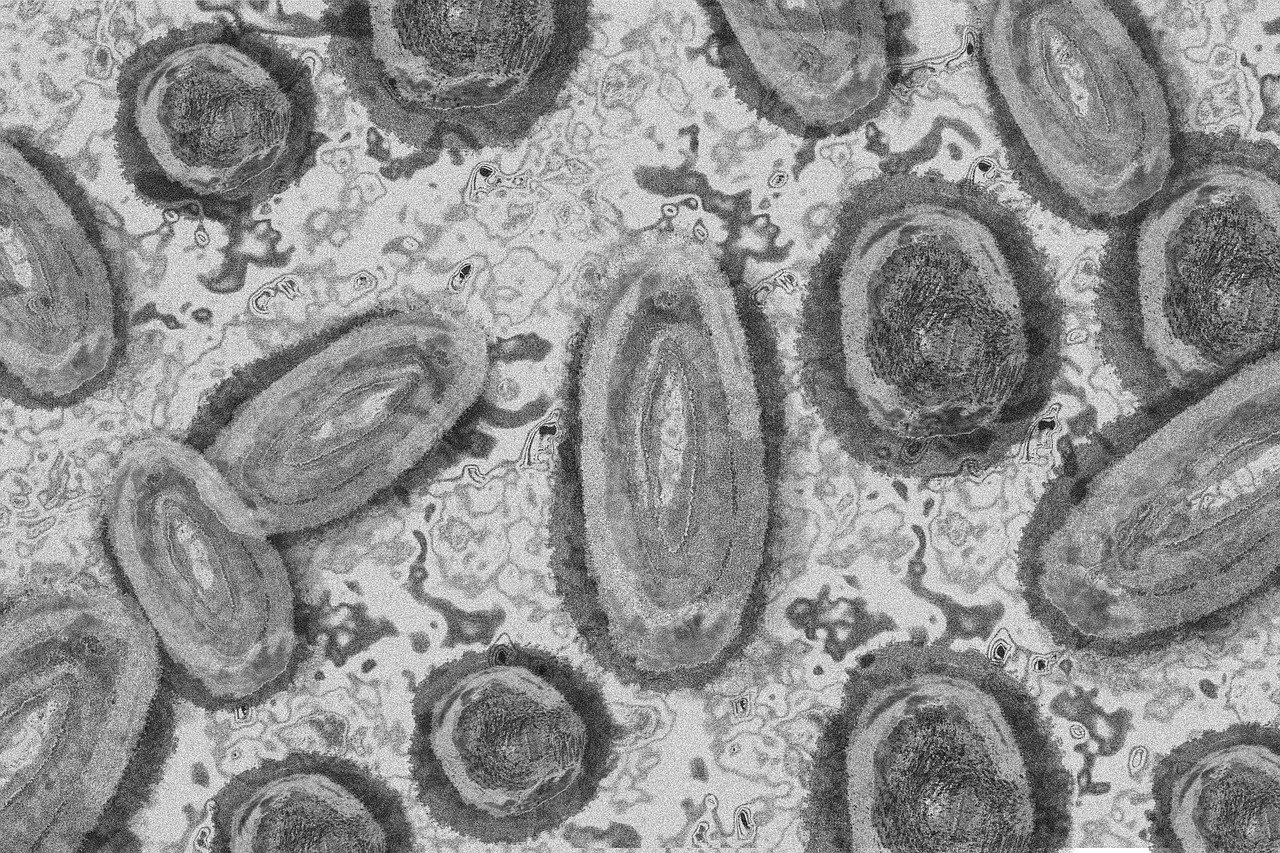
UK designates monkeypox as a notifiable disease
The UK Health Security Agency said monkeypox would be designated as a notifiable infectious disease from Wednesday, meaning doctors in England will have to notify local authorities when they suspect a patient has the virus. "Rapid diagnosis and reporting is the key to interrupting transmission and containing any further spread of Monkeypox," Wendi Shepherd, Monkeypox incident director at UKHSA, said in a statement.
Novavax COVID vaccine gets backing of U.S. FDA advisers
Advisers to the U.S. Food and Drug Administration on Tuesday voted to recommend authorization of Novavax Inc's COVID-19 vaccine for adults.
Novavax says COVID vaccine for U.S. to be manufactured by India's Serum
Novavax Inc said on Tuesday that its COVID-19 vaccine initially available in the United States, if authorized, will be manufactured by the Serum Institute of India. "So all vaccines that are being distributed globally commercially are being made in a single facility by our partners (the Serum Institute in India) that includes the vaccines which are being deployed around the world as well as the ones that will be initially deployed in the U.S.," said Chief Medical Officer Filip Dubovsky.
Novavax COVID vaccine backed for authorization by U.S. FDA panel
Advisers to the U.S. Food and Drug Administration on Tuesday voted overwhelmingly to recommend that the agency authorize Novavax Inc's COVID-19 vaccine for use in adults, which the drugmaker hopes can become the shot of choice among some American vaccine skeptics. The panel of outside vaccine experts voted 21-0 with one abstention in favor of the vaccine for those 18 and older.
Bluebird bio's blood disorder therapy effective - FDA staff
Bluebird bio's blood disorder treatment demonstrates "clinically meaningful" benefit in patients, staff reviewers at the U.S. Food and Drug Administration said in briefing documents published on Tuesday. The company has applied for approval of beti-cel as a one-time gene therapy for the treatment of Beta-thalassemia patients dependent on blood transfusions.
Bavarian Nordic eyes more monkeypox vaccine orders amid global "wake-up call"
Biotechnology company Bavarian Nordic expects to sign more contracts for its vaccine against monkeypox after "overwhelming" interest from governments around the world, its management told investors on Tuesday. The Danish company, which produces the only vaccine approved in the United States and Canada against monkeypox, has become the focus of a global rush from governments around the world hoping to bolster their preparedness against the virus.
(With inputs from agencies.)
- READ MORE ON:
- UKHSA
- The UK Health Security Agency
- Danish
- India
- U.S. FDA
- The U.S. Department of Justice
- U.S.
- The U.S. Centers for Disease Control and Prevention
- Monkeypox
- Centers for Disease Control and Prevention
- COVID
- Eli Lilly
- Bavarian Nordic
- England
- United States
- European Centre
- Novavax
- World Health Organization's
- U.S. Food and Drug Administration
- New Jersey
ALSO READ
Artemis II: A Cosmic Leap for Canada and U.S. Space Relations
Canada's Economic Pulse: Job Growth, Tariffs, and Unemployment in Focus
Canada's Job Market Faces Slow Recovery Amid Economic Challenges
Canada's Job Market: Slight Growth Amid Challenges
Canada's Firm Stance: Condemning Violence in the Middle East










